The standard of care for proliferative retinopathies, such as diabetic retinopathy and age-related macular degeneration, has long relied on invasive intravitreal injections. These direct-to-eye needle procedures are often painful and carry risks of infection or increased ocular pressure. However, a scientific breakthrough published in April 2026 by researchers at Wayne State University and Washington State University introduces a “smart” alternative: the trehalose-based glycodendrimer (Tre-D). This nanocarrier enables systemic drug delivery, allowing sight-saving medication to reach the retina via a simple body injection rather than an intraocular needle.
What’s New: Quantified Breakthroughs
- Targeted Delivery: The Tre-D nanocarrier is the first organic nanoparticle capable of localizing specifically to damaged retinal vessels after systemic injection, ignoring healthy vasculature.
- Elimination of Invasiveness: In murine models, the system successfully delivered Axitinib (a potent growth inhibitor) directly to neovascular tufts, reducing the need for direct eye injections.
- Smart Binding: The presence of trehalose allows the carrier to “recognize” and bind to aberrant tufts that block light and cause blindness.
- Source: Research led by Dr. Nikhlesh Singh (Wayne State University) and Dr. Anjali Sharma (Washington State University), published in Theranostics, April 2026.
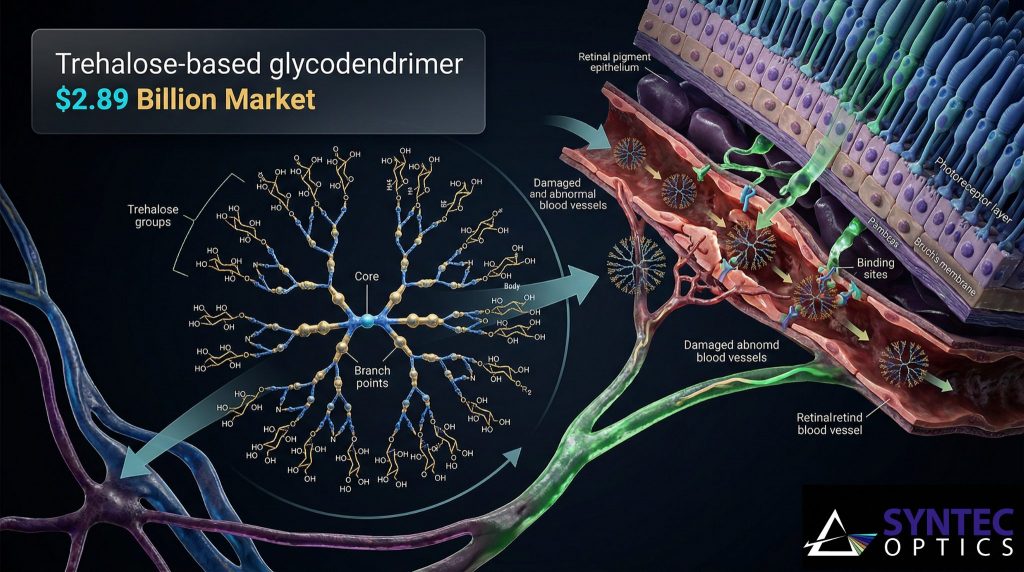
The Science: How It Works
The trehalose-based glycodendrimer functions as a molecular homing missile. When injected into the bloodstream, most drugs distribute throughout the body, often leading to toxicity or insufficient concentrations at the target site. The “Tre-D” structure utilizes the chemical properties of trehalose to identify the specific biochemical markers of damaged blood vessels in the eye.
Once the nanocarrier binds to these “tufts” in the retina, it releases its therapeutic payload. This targeted approach is critical because the drugs used to stop vessel growth are often toxic if they circulate freely. By encapsulating the drug within a glycodendrimer, researchers can ensure the medication only becomes active where it is needed most, preserving vision without the trauma of repeated eye surgery.
The Syntec Perspective: From Lab to Fab
As this non-invasive therapy moves toward clinical deployment, the demand for high-resolution ophthalmic imaging to monitor “tuft” reduction will skyrocket. Syntec Optics provides the manufacturing backbone for the hardware that makes this possible:
- Precision Housing & Barrels: Our ability to maintain tolerances of .0002–.0005 between cells and barrels is essential for the multi-element zoom systems used in next-generation fundus cameras and OCT systems that will track Tre-D efficacy.
- Polymer & Glass Integration: Syntec specializes in complex optical assemblies, including Asphere Barrels and Micro-Laser components, which are critical for the miniaturized diagnostic tools required in modern clinics.
- Advanced Metrology: We utilize leading-edge metrology to ensure every lens used in biomedical imaging meets the rigorous quality standards required for FDA-regulated medical devices.
Industry Applications & Deployment
This technology is positioned to disrupt the Ophthalmic Diagnostic Devices Market, which is projected to reach $2.89 billion in 2026 according to recent industry forecasts (Fortune Business Insights). The shift toward systemic delivery will create a massive need for portable, high-fidelity imaging adapters for smartphones and remote diagnostic tools.
North America continues to dominate this sector, holding over 38% of the global market share. We expect the first commercial deployments of Tre-D monitoring systems to appear in specialized ophthalmic clinics and hospital settings where high-precision diagnostics are mandatory.
Key Takeaways
- Shift in Care: Moving from invasive eye injections to systemic “flu-shot” style delivery via trehalose-based glycodendrimers.
- Enhanced Safety: Reduced risk of ocular infection and inflammation by avoiding direct needle contact with the eye.
- Manufacturing Readiness: Syntec Optics is ready to scale the optomechanical assemblies and precision coatings needed for the imaging systems that will validate these treatments.
References
- Wayne State University: “Research team develop nanocarrier that may offer new, less-invasive treatment option for retinopathy” – https://today.wayne.edu/medicine/news/2026/04/08/research-team-develop-nanocarrier-that-may-offer-new-less-invasive-treatment-option-for-retinopathy-68812
- Fortune Business Insights: “Ophthalmic Diagnostic Devices Market Size, Share | Report 2034” – https://www.fortunebusinessinsights.com/ophthalmic-diagnostic-devices-market-102184